You should know that kratom lacks FDA approval and carries serious health risks you’re unlikely to hear from vendors. Unregulated products may contain contaminants like lead, bacteria, and undeclared synthetic opioids. You face potential neurological complications, liver damage, respiratory depression, and dependency with chronic use. Clinical evidence supporting kratom’s safety remains sparse. Withdrawal symptoms can be intense. Understanding these dangers fully requires examining what specific complications emerge across different users and situations.
Lack of FDA Approval and Unproven Medical Benefits

Although kratom products are widely marketed for pain, anxiety, opioid withdrawal, and other conditions, they haven’t received FDA approval for any medical use. This absence of approval reflects a critical gap: you won’t find FDA-reviewed evidence supporting kratom’s safety or effectiveness at specific doses for any condition.
The clinical research supporting kratom remains sparse and weak. Most studies rely on animal data, case reports, or uncontrolled surveys rather than rigorous human trials. You’re facing therapeutic uncertainty because long-term safety data don’t exist, leaving unknown risks about chronic use and drug interactions. Additionally, kratom products are unregulated and may contain metals, bacteria, or other harmful substances that pose contamination risks. The FDA has funded limited human studies, including a single ascending dose study and a human abuse potential study awarded in September 2024, but results remain pending and insufficient to establish safety standards.
Companies making unlawful marketing claims about kratom’s health benefits operate outside regulatory standards. In response to these concerns, the FDA issued warning letters to 7 companies for illegally distributing related products and published recommendations about potential health risks. Until adequate evidence undergoes FDA review, you should recognize that kratom products exist in a regulatory gray zone where therapeutic benefit remains unproven and safety unverified.
Contamination and Product Variability Concerns
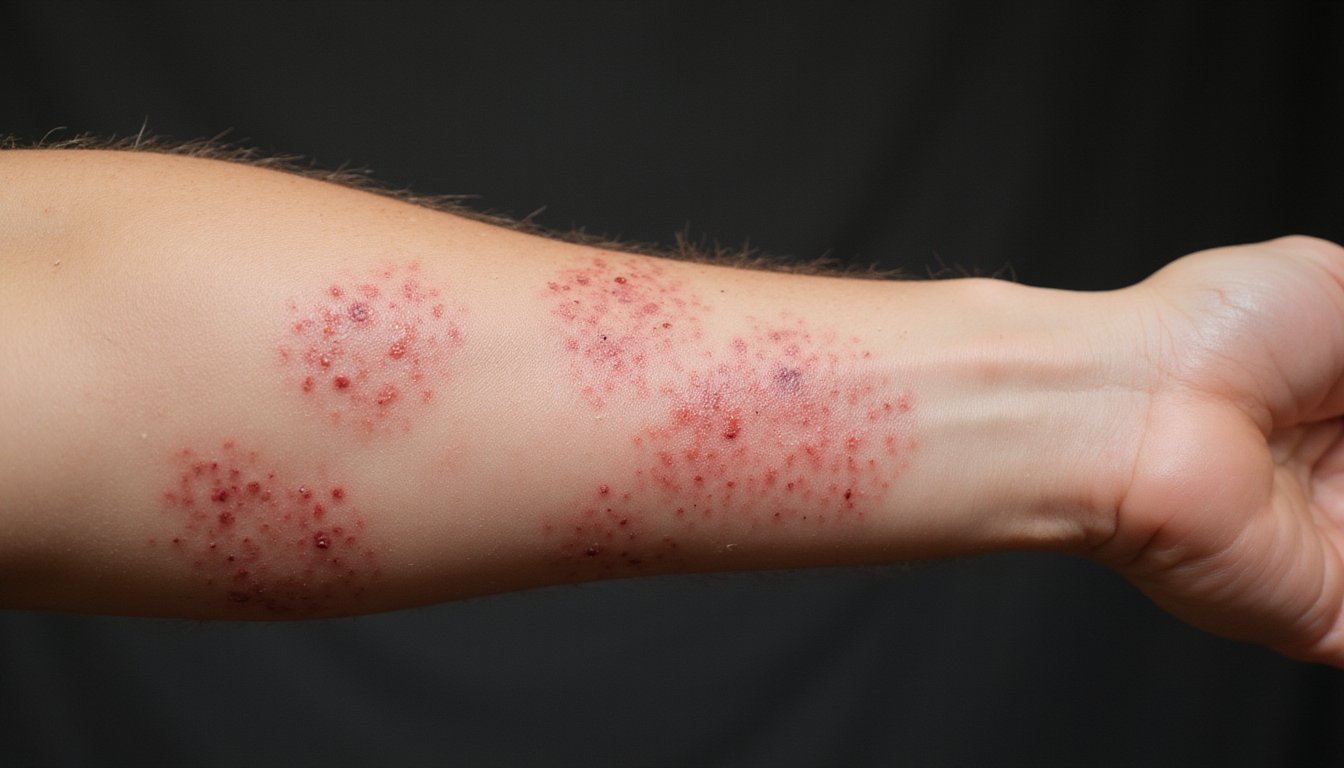
Because kratom products aren’t subject to FDA manufacturing standards, you’re exposed to multiple contamination risks that regulated pharmaceuticals and dietary supplements don’t pose. Microbial contamination risks include Salmonella outbreaks linked to kratom, affecting 41 U.S. states. You face toxic impurities concerns from heavy metals like lead and nickel, which accumulate through chronic use without mandatory limits on content.
| Contamination Type | Source | Health Risk |
|---|---|---|
| Salmonella & pathogens | Unregistered producers | Foodborne illness outbreaks |
| Lead, nickel | Cultivation soils | Neurologic, renal toxicity |
| Synthetic opioids | Undeclared adulterants | Overdose, unpredictable effects |
| Alkaloid variability | Batch differences | Inconsistent potency |
Unregulated manufacturers add pharmaceuticals and research chemicals without label disclosure. Fragmented supply chains complicate traceback investigations when contaminated batches cause harm. Third-party testing remains inconsistent, enabling adulteration and misbranding across online vendors and retail outlets. The rising number of Poison Control Center calls related to kratom demonstrates the growing health risks associated with unregulated product availability. Since kratom compounds vary widely depending on dosage, potency, and formulation, contaminants in unregulated products can produce unpredictable effects beyond the substance’s intended alkaloid profile. Additionally, psychological and physiological dependence on kratom can develop through regular use, compounding the risks posed by contaminated or variable products.
Acute Side Effects and Toxicity Risks

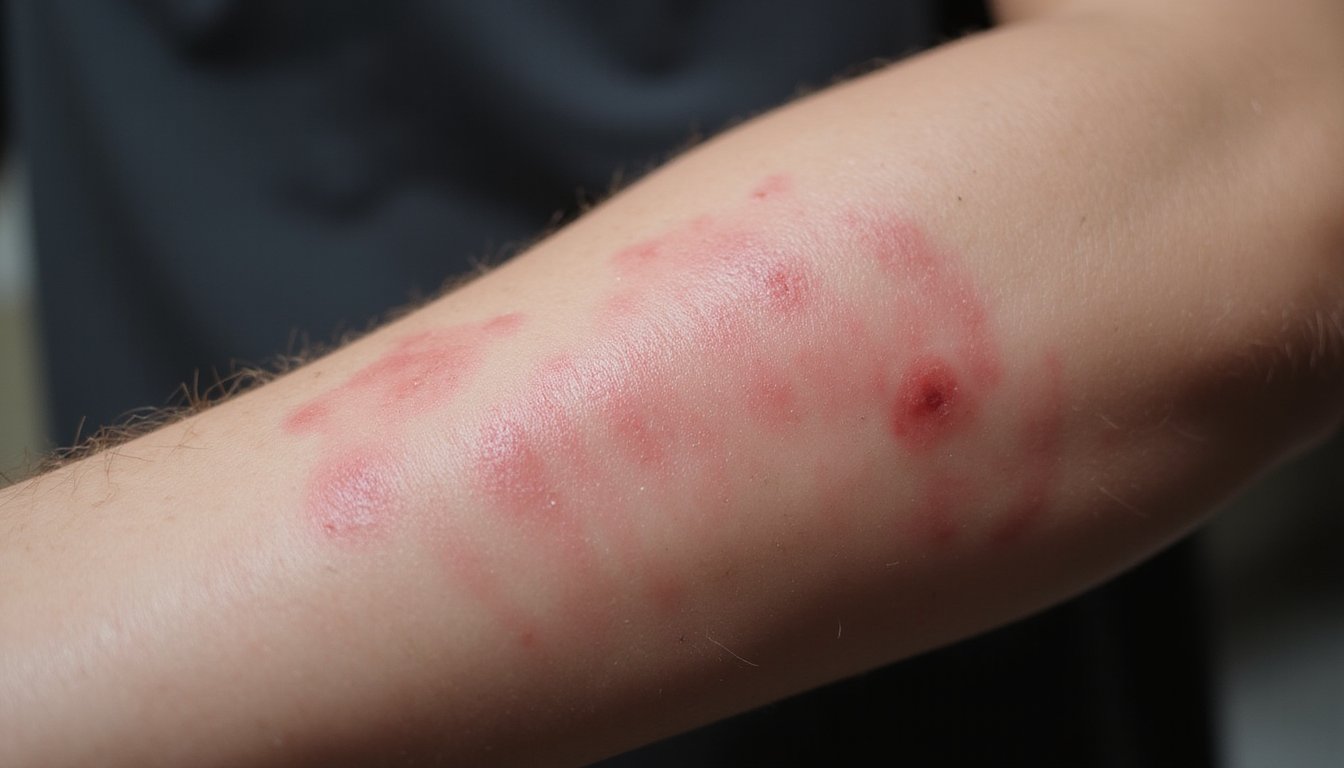
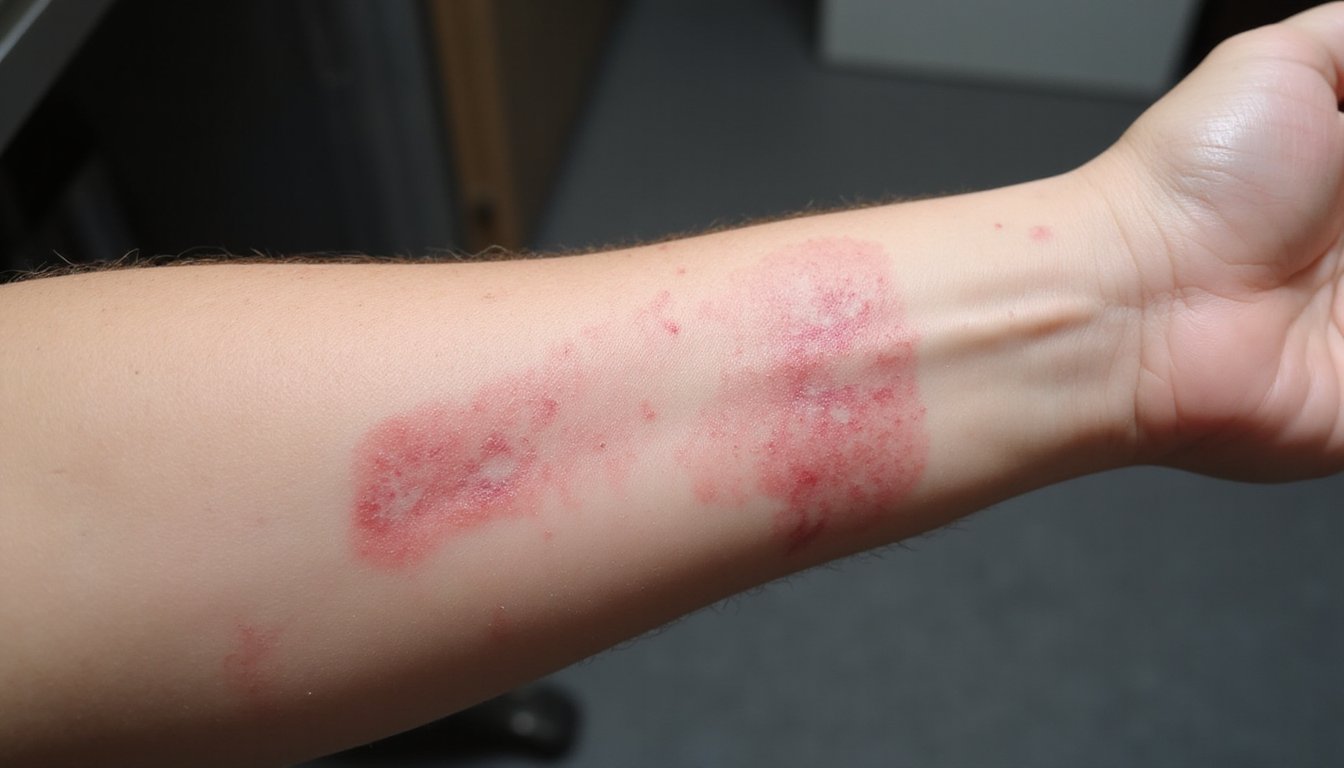
While kratom’s contamination risks create long-term health hazards, non-FDA-approved kratom also poses immediate dangers through acute toxicity that can manifest within hours of consumption. You may experience neurological complications including seizures, agitation, confusion, and altered mental states. The FDA and DEA specifically warn of seizure risk, with 12 confirmed cases among survivors. Cardiotoxic effects threaten your cardiovascular system through tachycardia, heightened blood pressure, and increased heart rate. Pulmonary edema occurs in 74.3% of fatal cases, causing dangerous fluid accumulation in your lungs. You might also develop respiratory depression, shallow breathing, and aspiration risks. Hepatotoxicity presents additional concern, with documented cases showing raised liver enzymes and cholestasis. These acute effects can progress rapidly to loss of consciousness, requiring immediate emergency intervention. Additionally, kratom products may be adulterated with more active ingredients than naturally present, which can intensify the severity and unpredictability of these acute toxic reactions.
Dependence, Addiction, and Withdrawal Symptoms
You can develop tolerance to kratom with regular use, requiring progressively higher doses to achieve the same effects, a hallmark of physical dependence. When you stop using kratom after sustained consumption, you’ll likely experience withdrawal symptoms ranging from muscle aches and insomnia to anxiety and intense cravings that can persist for weeks. The acute withdrawal phase, typically occurring 1 to 3 days after your last dose, brings the highest intensity of physical and emotional symptoms including nausea, vomiting, sleeplessness, and depression. These withdrawal effects, combined with your continued use despite negative consequences, indicate that non-FDA-approved kratom poses significant risks for addiction and long-term physiological disruption.
Regular kratom use produces tolerance, a hallmark feature of opioid-like substances, requiring escalating doses to maintain the same effects, particularly with frequent dosing patterns. You develop physical dependence through kratom’s partial activation of mu-opioid receptors, driving compulsive use behaviors and withdrawal symptoms upon cessation.
| Risk Factor | Clinical Outcome | Dependence Severity |
|---|---|---|
| >5g per serving | Reduced analgesic effects | Moderate-High |
| >3 times daily | Mood escalation tolerance | High |
| Daily use | Withdrawal upon cessation | Severe |
| Dose escalation patterns | Amplified alkaloid exposure | Critical |
Mitragynine pharmacokinetics intensify opioid-like risks as you increase consumption. You’ll experience tolerance manifesting as diminished euphoria and sedation, forcing higher doses to “feel normal” rather than achieve initial desired effects. This cycle increases your exposure to active alkaloids, substantially elevating physical dependence risk, particularly problematic given kratom’s unregulated status and lack of FDA oversight. Research shows that dose frequency rather than dose amount is more strongly associated with withdrawal and kratom use disorder symptoms, suggesting that how often you use kratom may be a critical factor in developing dependence.
Withdrawal Symptoms and Long-Term Effects
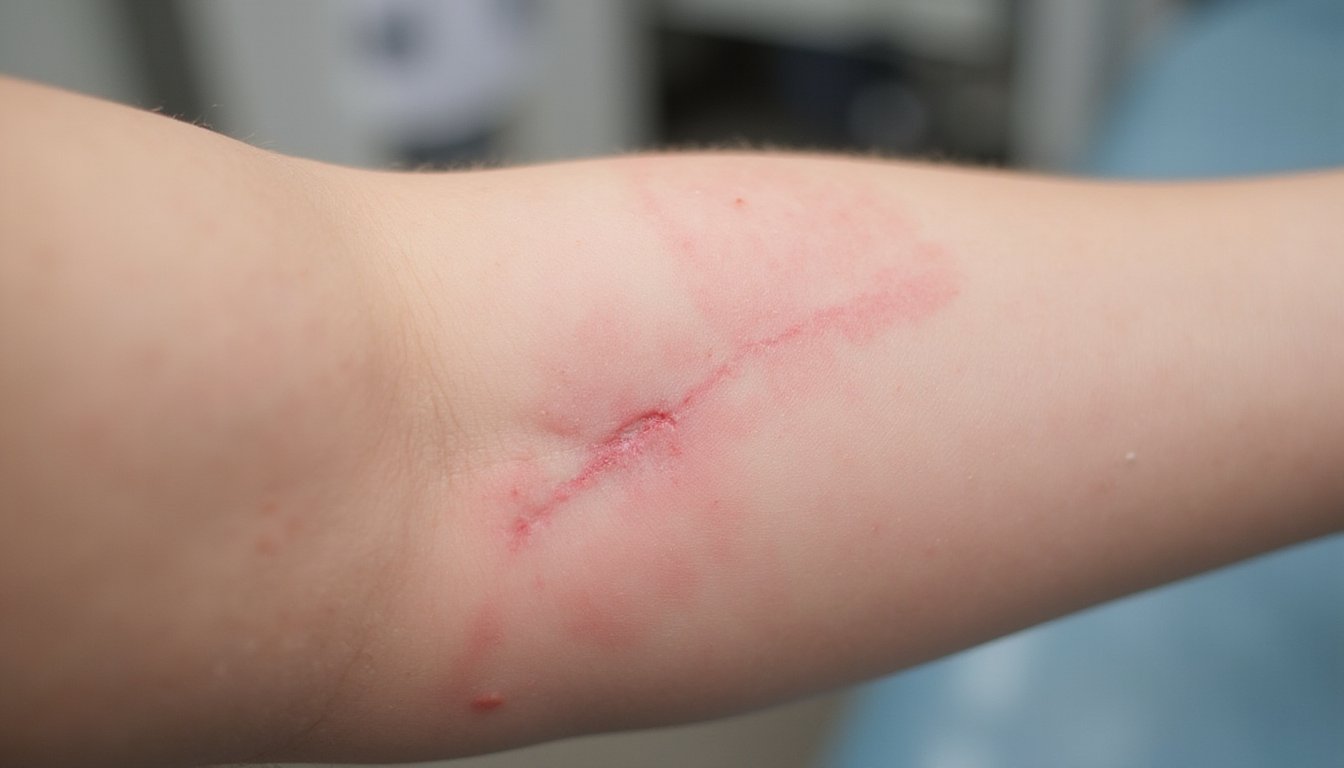
As your body adapts to kratom’s alkaloid exposure, cessation triggers a distinct withdrawal syndrome characterized by depressed mood, irritability, muscle aches, anxiety, and diarrhea, symptoms that typically emerge 12, 48 hours after your last dose and persist for 1, 7 days. While generally milder than opioid withdrawal, these symptoms remain moderately severe and often self-manageable.
Long-term kratom use carries significant consequences. Approximately 29.5% of users meet Kratom Use Disorder criteria, with 8.5% experiencing severe addiction. Extended use correlates with mental health disorder onset, polysubstance abuse patterns, and occupational/familial disruption. Common withdrawal symptoms include gastrointestinal upset, restlessness, and craving, which emerge as the body’s physical dependence manifests during discontinuation. Research indicates that gradual dose tapering and non-opioid approaches may provide relief during kratom withdrawal management. Symptom mitigation strategies and cognitive behavioral interventions show promise for managing withdrawal and preventing relapse. However, 80% of abusers require professional assistance for successful cessation, emphasizing kratom’s substantial addiction potential despite its non-FDA-approved status.
Organ-Specific Health Complications

Because kratom isn’t FDA-approved, its consumption poses documented risks across multiple organ systems without established safety standards or quality controls. Your liver faces serious threats from kratom exposure, with hepatotoxicity cases requiring acute liver failure interventions and liver transplant needs. Cardiovascular complications including hypertension and tachycardia appear in thousands of poison center reports. You’re vulnerable to respiratory depression resembling opioid effects, potentially causing breathing suppression during overdose. Gastrointestinal damage ranges from nausea and constipation to severe salmonella infections that claimed over 35 lives. Neurological effects include seizures, tremors, and confusion documented across adverse event logs. Additionally, an estimated 1.9 million Americans have reported using kratom in the previous year despite these serious health risks. Product contaminants amplify these organ-specific injuries substantially. Without FDA oversight, you cannot verify kratom’s purity, potency, or safety profile before consumption.
Drug Interactions and Vulnerable Populations
Without FDA oversight, kratom’s pharmacological interactions with prescription medications create substantial risks you must understand before consumption.
| Drug Class | Interaction Mechanism | Clinical Risk |
|---|---|---|
| CYP3A4/2D6 substrates | Enzyme inhibition slows metabolism | Increased drug concentrations, toxicity |
| CNS depressants | Additive respiratory depression | Severe sedation, breathing compromise |
| Serotonergic agents | Excess serotonin accumulation | Syndrome, seizures, cardiac effects |
You face heightened vulnerability if you’re pregnant, breastfeeding, or taking multiple CYP-metabolized medications. Ethnicity-based metabolism variations further complicate individual risk prediction. Benzodiazepines, opioids, SSRIs, and antipsychotics warrant particular caution. Naltrexone co-use precipitates withdrawal syndrome. Kratom toxicity symptoms have been documented at doses exceeding 8 g/day, and when combined with long-term use patterns, the risk of dependence and tolerance substantially increases your vulnerability to adverse outcomes. Without standardized kratom alkaloid content or FDA regulation, you cannot predict interaction severity or onset timing, making combination therapy inherently dangerous regardless of dosing adjustments. Mitragynine and O-desmethyltramadol are the primary alkaloids in kratom that interact with hepatic enzyme systems to create unpredictable metabolic outcomes.
Searching for answers about whether kratom is FDA-approved can feel like the beginning of a much harder realization, one that leaves you questioning every dose you took believing it was safe. At medical detox in tampa fl, we understand how unsettling it is to discover that something you trusted has never been approved, tested, or deemed safe by any regulatory authority, and that its grip may already be stronger than you realized. We connect you with reputable Kratom Detox Programs and treatment centers staffed by professionals who know exactly what it takes to help you find your way back. You don’t have to navigate this alone. Call 740-562-7398 today and let us help you take that first step toward healing.
Frequently Asked Questions
How Can I Safely Stop Using Kratom if I’ve Developed Dependence or Addiction?
You should taper dosage carefully under medical supervision rather than quitting abruptly, as sudden cessation causes severe withdrawal symptoms like anxiety, insomnia, and muscle aches. Seek medical support to develop a structured plan reducing your dose by 5, 20% every few days. Your doctor can prescribe symptomatic medications for discomfort and screen for organ damage. Combine tapering with behavioral therapy, coping strategies, and peer support to reduce relapse risk effectively.
What Should I Do if I Experience a Kratom Overdose or Severe Adverse Reaction?
If you’re experiencing a kratom overdose, you’ll need to seek emergency medical care immediately by calling 911. Contact poison control for specialized toxicological guidance. While awaiting paramedics, monitor your critical signs and note the amount you’ve ingested. Don’t induce vomiting. Medical professionals will provide decontamination through activated charcoal or gastric lavage, manage cardiovascular symptoms, and treat seizures if they develop. Prompt intervention drastically reduces overdose complications.
Are There Legal Alternatives to Kratom for Managing Pain or Mood Symptoms?
You’ve got several evidence-based options. FDA-approved medications like gabapentin, pregabalin, and duloxetine effectively manage neuropathic and musculoskeletal pain. For mood support, tricyclic antidepressants offer clinical benefit. You can also explore natural supplements, turmeric, ginger, and valerian root, which provide anti-inflammatory and anxiolytic properties without regulatory concerns. CBD oil presents another alternative therapy with documented pain-relief benefits. These alternatives carry established safety profiles, unlike unregulated kratom products.
How Long Does Kratom Remain in My System After Stopping Use?
You’ll typically detect kratom in your urine for 3, 7 days after stopping use, though heavy users may experience extended detection windows. Mitragynine’s half-life averages 6.6 hours, meaning multiple half-lives pass before complete elimination. Your withdrawal timeline depends on usage frequency and dosage, daily users often experience prolonged clearance. Individual factors like metabolism, kidney function, and body composition considerably influence how long kratom persists in your system.
What Testing or Certifications Should I Look for When Purchasing Kratom Products?
You should verify that your kratom vendor maintains AKA GMP Certification and provides Certificates of Analysis from ISO/IEC 17025:2017-accredited labs. Guarantee product batch testing matches your specific lot, with results dated within six months. Third party laboratory analysis should document mitragynine content, identity verification, and microbiological pathogen screening. Request documentation of independent annual audits. These certifications demonstrate rigorous quality control and reduce contamination risks before you purchase.